Main Energy Levels or Shells, Sublevels or Subshells
Shells and subshells
Electron configuration was first conceived of under the Bohr model of the atom, and it is still common to speak of shells and subshells despite the advances in understanding of the quantum-mechanical nature of electrons.
An electron shell is the set of allowed states, which share the same principal quantum number, n (the number before the letter in the orbital label), that electrons may occupy. An atom's nth electron shell can accommodate 2n2 electrons, e.g. the first shell can accommodate 2 electrons, the second shell 8 electrons, and the third shell 18 electrons. The factor of two arises because the allowed states are doubled due to electron spin—each atomic orbital admits up to two otherwise identical electrons with opposite spin, one with a spin +1/2 (usually noted by an up-arrow) and one with a spin −1/2 (with a down-arrow).
A subshell is the set of states defined by a common azimuthal quantum number, ℓ, within a shell. The values ℓ = 0, 1, 2, 3 correspond to the s, p, d, and f labels, respectively. The maximum number of electrons that can be placed in a subshell is given by 2(2ℓ + 1). This gives two electrons in an s subshell, six electrons in a p subshell, ten electrons in a d subshell and fourteen electrons in an f subshell.
The numbers of electrons that can occupy each shell and each subshell arises from the equations of quantum mechanics, in particular the Pauli exclusion principle, which states that no two electrons in the same atom can have the same values of the four quantum numbers.
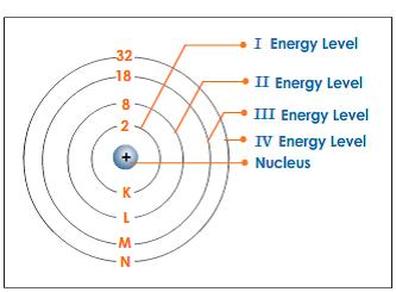
Maximum number of electrons that can be accommodated in a shell is given by 2n2 where n = shell number
- For 1st energy level, n = 1Maximum number of electrons in 1st energy level = 2n22 x (1) 2 = 2
- For 2nd energy level n = 2Maximum number of electrons in the 2nd energy level = 2n22 x 22 = 2 x 4 = 8
- For 3rd energy level n = 3Maximum number of electrons in the 3rd energy level = 2n2= 2x(3) 2= 2 x 9 = 18
- For 4th energy level n = 4Maximum number of electrons in the 4th energy level = 2n2= 2x(4) 2= 2x16 = 32
The outermost shell of an atom cannot accommodate more than 8 electrons, even if it has a capacity to accommodate more electrons. This is a very important rule and is also called the Octet rule. The presence of 8 electrons in the outermost shell makes the atom very stable.